Abstract
Our unit aims to understand how coral reef fish adapt to environmental changes such as climate change, heatwaves, overfishing and urbanization. Earth's oceans are warming and acidifying due to increasing anthropogenic CO2 production, and extreme events such as marine heatwaves are increasing in frequency, duration, and magnitude. Coral reef fish are especially vulnerable to spikes in temperature because they are already adapted to living close to their thermal limits. Determining which species can adapt to rapid environmental change, and how they will do so, is critical for predicting the ecological effects of climate change and their impact on the world economy.
In our laboratory research, we investigate how short-term as well as heritable changes in gene expression in multiple fish species are affected by increases in sea level temperature and acidity. We study animals that are descended from wild breeding pairs and have been reared in our aquariums over several generations. Using a unique device called the Heatwaves Simulator aquarium system based at OIST’s Marine Science Station, we expose fish to specific conditions, including the temperature and acidity level predicted to occur in seawater by the end of the century. We combine these manipulations with a range of advanced genomic technologies to identify the cellular mechanisms that allow coral reef fish communities in Okinawa and around the world to acclimate and adapt to changing conditions.
1. Staff
Tae Woo Ryu, Research Unit Group Leader
Roger Huerlimann, Postdoctoral Scholar
Shannon McMahon, Postdoctoral Scholar
David Miller, Visiting Researcher
Jamila Rodrigues, Visiting Researcher
Emanuela Borgnino, JSPS fellow
Nicolas Dierckxsens, OIST Interdisciplinary Postdoctoral Scholar (50/50 with Nick Luscombe Unit)
Erina Kawai, Research Unit Scientific Diver
Jeffrey Jolly, Research Unit Technician
Chengze Li, Research Unit Technician
Gelyn Bourguignon, Fish Husbandry Technician
Michael Izumiyama, Graduate Student
Billy Moore, Graduate Student
Callum Hudson, Graduate Student
Ayşe Haruka Oshima Açıkbaş, Graduate Student
Kaisar Dauyey, Visiting Research Student
Johanna Linnea Johansson, Research Intern
Yoko Shintani, Research Unit Administrator
2. Collaborations
2.1 Anemonefish development and genomics
Type of collaboration: Scientific Collaboration
Researcher:
Professor Vincent Laudet, OIST, Japan
2.2 Coral Reef fish adaptation to marine heatwaves
Type of collaboration: Scientific Collaboration
Researchers:
Professor Philip Munday, James Cook University, Australia
Professor Jennifer Donelson, James Cook University, Australia
Professor Jacob Johansen, University of Hawaii
2.3 Coral Reef fish adaptation to Ocean Acidification
Type of collaboration: Scientific Collaboration
Researcher:
Professor Celia Schunter, The University of Hong Kong, HongKong
2.4 Coral Reef fish genomics
Type of collaboration: Scientific Collaboration
Researchers:
Deputy Director Piero Carninci, RIKEN Center for Integrative Medical Sciences, Japan
Professor Jesper Tegnér, KAUST, Saudi Arabia
Professor Valerio Orlando, KAUST, Saudi Arabia
2.5 Coral Reef fish Physiology
Type of collaboration: Scientific Collaboration
Researcher:
Professor Martin Grossel, University of Miami, USA
2.6 Impact of coastal development on coral reef ecosystems
Type of collaboration: Scientific Collaboration
Researchers:
Professor James Davis Reimer, University of Ryukyus, Japan
Professor Noriyuki Satoh, OIST, Japan
2.7 Natural analogues of climate stressors
Type of collaboration: Scientific Collaboration
Researcher:
Dr. Riccardo Rodolfo-Metalpha, UMR ENTROPIE, Nouvelle Calédonie
2.8 Natural Volcanic CO2 Analogues
Type of collaboration: Scientific Collaboration
Researchers:
Professor Ivan Nagelkerken, The University of Adelaide, Australia
Professor Sylvain Agostini, University of Tsukuba, Japan
2.9 Okinawa Coral spawning and development
Type of collaboration: Scientific Collaboration
Researcher:
Professor David J. Miller, James Cook University, Australia
2.10 Sharks physiology under climate stressors
Type of collaboration: Scientific Collaboration
Researchers:
Professor Jodie L. Rummer, James Cook University, Australia
Professor Rui Rosa, Faculdade de Ciências da Universidade de Lisboa, Portugal
2.11 Transgenerational acclimation
Type of collaboration: Scientific Collaboration
Researcher:
Professor Jennifer Donelson, James Cook University, Australia
2.12 Responses of Sea Urchins to Climate Change
Type of collaboration: Scientific Collaboration
Researchers:
Dr. Ben Harvey, University of Tsukuba, Japan
Prof. Bayden Russell, University of Hong Kong, Hong Kong
Professor Valerio Orlando, KAUST, Saudi Arabia
2.13 Microbiome of Commercially Important Sea Urchins
Type of collaboration: Scientific Collaboration
Okinawa Prefectural Sea Farming Center
2.14 Biology of Endosymbiotic Sponge-Dwelling Polychaetes (Haplosyllis spp.)
Type of collaboration: Scientific Collaboration
Researchers:
Chloé Loïs Fourreau, University of the Ryukyus, Japan
Professor James Reimer, University of the Ryukyus, Japan
2.15 Microplastic Identification and Characterization (I’m sure you already planned to include this, but just in case)
Type of collaboration: Scientific Collaboration
Researchers:
Dr. Samantha Phan, OIST, Japan
Professor Jesper Tegnér, KAUST, Saudi Arabia
Professor Christine Luscombe, OIST, Japan
3. Activities and Findings
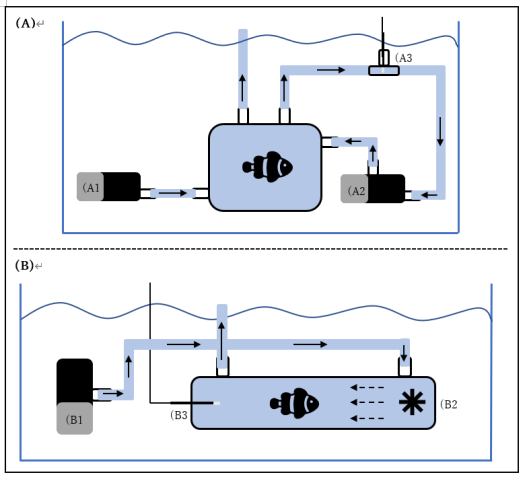
3.1 Design and Construction of the OIST Heatwaves Simulator aquariums system.
This year our unit in collaboration with Luxaqua, we designed and constructed the OIST Heatwaves Simulator, a unique aquariums system that will allows us and our collaborators to recreate a future environment including the simulation of marine heatwaves at the OIST Marine Science Station.
The OIST Heatwaves Simulator consist of three separate tank systems (Figure 1); 1) 10, 100L outdoor breeding pair tanks that will be used to house brood stock breeding pairs, 2) 10, 75L larval rearing tanks, that from 2022 onwards (F2, F3) will be used in the larval rearing stage of this multi-generational experiment, 3) 144, 55L tanks that will house experimental juvenile fish in different treatments. Temperature within these experimental tanks is controlled by four inline heaters, that allow five different temperature treatments to be specified in each individual tank. Temperature is controlled and monitored by automated software and associated temperature probes. Software also has remote monitoring and control capabilities.
This system is unique in the world and it will help to incentive collaborators from Japan and overseas to come to OIST and work with us at the marine station.
3.2 Coral Reef fish Physiology and Metabolic Performance
To assess the effects of the different heatwave treatments on coral reef fish’s metabolic rates, in collaboration with Loligo Systems, we set up at the OIST Marine Station a 170ml mini-swim tunnel (Figure 1). Water flow within the swim tunnel is generated by a propeller, and laminar flow is created by a series of flow straightening plastic honeycomb meshes. The velocity of water within the tunnel is controlled by Loligo Systems AutoResp software and is calibrated using Loligo Systems fluorescent PE microspheres and the associated Digital Particle Tracking Velocity software. An oxygen dipping probe is inserted into the swim tunnel, allowing for continuous measurements of oxygen concentration within the tunnel. A pump can be used to flush the swim tunnel with fresh seawater from the waterbath if required. The swim tunnel is submerged within a temperature controlled waterbath allowing the temperature within the swim tunnel to be manipulated. Flushing of the swim-tunnel and resting chambers, monitoring of oxygen concentrations and temperature, and calculations of oxygen consumption rates are all completed automatically by Loligo Systems AutoResp software.
3.3 Amphiprion ocellaris Spawning and Larval Rearing
In order to conduct the planned long-term multi-generational experiment detailed above, we first needed to ensure that wild-caught A.ocellaris from Okinawan coral reef systems would spawn within aquaria. To test this, one breeding pair of A.ocellaris was acquired from local fishermen in September 2019. This pair was placed within a 200L aquaria at OIST Marine Science Station with a sea anemone (Heteractis magnifica) and multiple rocks.
On the 31st of May 2020 eggs were observed within the breeding pair tank (Figure 1). The development of eggs was tracked using visual observation and comparisons to know stages of embryonic development. On the 7th of June, eggs were transferred to a larval rearing tank for hatching (Figure 3) and larval A.ocellaris were present in the tank on the 8th of June.
The successful spawning and larval rearing program of 2020 allowed us to develop for the first time at OIST Marine Science Station a larval rearing protocol.
3.4 The Sequencing of Amphiprion ocellaris, Amphiprion clarkii and Epinephelus malabaricus genomes.
Our unit sequenced the genomes of three coral reef fish from the water of Okinawa island, the Amphiprion ocellaris, Amphiprion clarkii and Epinephelus malabaricus (Table 1). All these genomes have been sequenced using PacBio long-reads sequencing combined with Illumina gDNA and transcriptome sequencing and binned into chromosomes using Hi-Seq contact map technology.
These genomes are among the most completed fish genomes ever sequenced and are available free for the entire community.
3.5 Natural Analogues of Future Climate
Unique sites which are natural analogues of future ocean have a community that already exists under this naturally occurring extreme environment and can be utilized as a natural laboratory. These sites are subjected to daily changes in ocean chemistry, have dynamic interactions between habitat, predator, and prey, and have existed in long timescales. Inhabitants may have also adapted to these extreme conditions through generations. Although conditions at each extreme site vary and are not perfect simulations of future oceans, they can provide a window into how species may respond to climate change.
Our unit successful performed field expeditions at the following natural analogues.
Bourake, New Caledonia
In the Autumn of 2023, in collaboration with IRD, the University of Tsukuba, and the Victoria University of Wellington, PhD Candidate Callum Hudson conducted a week-long expedition to Bourake, New Caledonia, where he collected urchin specimen (Echinometra spp.) from within the acidified and warmed tidal mangrove lagoon, and nearby reference reef sites.
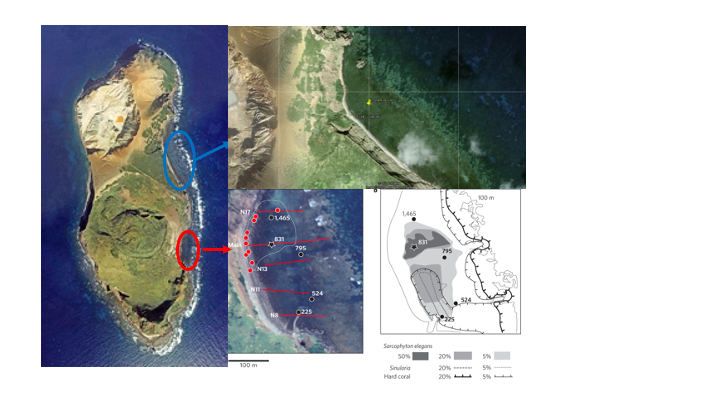
Ioutorijima, Japan
In the fall of 2020, our team and collaborators from Tokyo University and the University of the Ryukyus conducted a three-day trip to the uninhabited island Ioutorijima. We collected eDNA samples and placed baited remote underwater cameras (BRUV) at the CO2 seep. We collected eDNA samples from the seep (future) and a nearby reef (control). We placed BRUVs with krill to determine the species that were present at the seep. Additionally, this trip produced a publication led by Reimer et al. (2021), which discovered and described a rare occurrence of aragonite-forming Nanipora at the CO2 seep site.
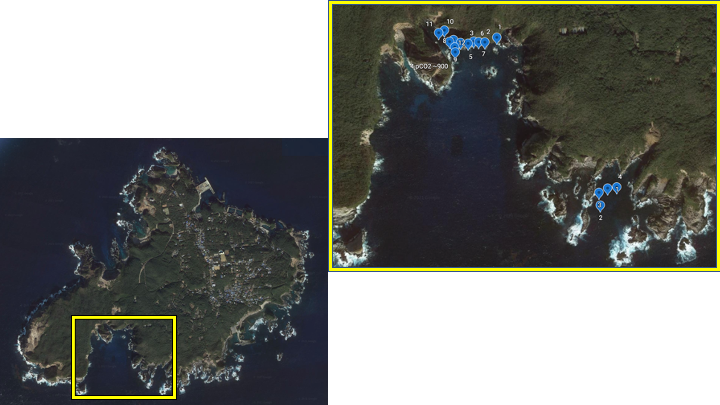
Shikine island, Japan
In the Summer of 2023, in collaboration with the University of Tsukuba, and the University of Adelaide, PhD Candidate Callum Hudson completed a week-long trip to Shikine-jima, Japan, to collect sea urchins (Echinometra spp.) from the CO2 seep. He performed respirometry and dissected key tissues crucial to echinoderm homeostasis in order to understand how urchins living at the seep site respond to long-term acidification exposure.
The third image, Figure 3, is a single image with the caption below it.
3.6 Tropicalizing Range-Shifts in the Japanese Coastlines via Environmental DNA Metabarcoding (eDNA).
Since the industrial age, anthropogenic activities have resulted in a consistent increase of sea surface temperature and ocean acidification, causing a world-wide shuffling of species, as part of an overall poleward range shift in both terrestrial and aquatic ecosystems. Specifically for coral reef recruitment, a tropical decrease and subtropical increase at the 20° latitude have been recorded with shifts to the more temperate areas of Southern Japan. This “tropicalization” raises the questions whether the Ryukyu arc islands could be experiencing this poleward shift along its axis (taking into account the effect of the Kuroshio current) and if it is also acting as a refugia for the migrating coral reef communities from near the equator.
Are the high latitude coral communities in Okinawa and Southern Japan the future areas of refuge for the range-shifting reef organisms, with the help of Kuroshio? Even if so, how would all the coastal development taking place in Okinawa affect this in the short-term and how immediately disruptive it could be? Could these migrating organisms be fitter compared to the original residents and what implications does this have for the future biodiversity of Okinawa? And how could the climate change be affecting the speciation process in the long-term, when compared to the past drastic changing phases in the climate?
The current climate-driven range shifts and therefore the opening-up of novel niches could serve as a window to see into and characterize the process of speciation with hybridization and adaptive radiation. Searching for and studying these newly created potential hybrid zones, we could gain a better understanding of speciation mechanisms in a changing and connected ecosystem. This hybrid swarm theory of adaptive radiation could be tested using population genetics and transcriptomics, in the nature and in the laboratory using the clownfish model organism.
In an increasingly warm world, it is crucial to understand how the changing conditions are affecting the species for more effective mitigation and conservation efforts. Effective design and management of MPAs can be achieved using the information gained from population connectivity and genomics studies, which can point to important sites of refuge/recruitment/nurseries. Temporal and spatial sampling for and studying eDNA across Okinawa and Japan can characterize the effects of the immediately disruptive anthropogenic activities such as coastal development and reconfiguration on biodiversity.
3.7 Microbiome of Commercially Important, locally threatened, Sea Urchins
Our unit has extended our collaboration with the Okinawa Prefectural Sea Farming Center to examine the gut-associated microbiome of cultured, commercially important, collector sea urchins (Tripneustes gratilla). The species is protected in Okinawa due to overfishing, however permission was granted by the Onna-village fishery for our team to collect a small number of individuals from reefs close to OIST. The microbiomes of cultured vs. wild individuals will be sequenced and compared in order to understand differences associated with urchin farming conditions that could have implications for sea urchin health and uni production.
3.8 The effect of suspended sediments on coral reef fish assemblages.
To date, there is a paucity of research on how near shore coral reef fish will be impacted by increased suspended sediments. This is especially true of coral reefs on larger islands such as those in the Coral Triangle, Hawaii, and Okinawa. To address this knowledge gap field surveys will be conducted at coral reef sites around Okinawa to properly access of suspended sediment events effect coral reef fish assemblages. Using historical data potential sites around Okinawa will be categorized by their suspended sediment history into “uncommon”, “common”, and “Chronic”. Three sites from each category will be identified and sampled from for this experiment. Surveys will be conducted before, during, and after increased suspended sediment events. The surveys will be conducted both by visual transects (Samoilys & Carlos, 2000) and by eDNA and eRNA sampling (Beng & Corlett, 2020). Using these two techniques will allow us a comprehensive assessment of the fish assemblage and provide us with a detailed understanding of how fish assemblage changes during these events. This knowledge will allow us and fellow researchers to more efficiently direct future research on suspended sediments towards the more sensitive and important coral reef fish species.
The effects of suspended sediment on the physiology and behaviour of marine fish are poorly understood. To improve our understanding of how suspended sediments may impact important marine habitats we will build a system at OIST Marine Science Station to allow the manipulation of suspended sediment in seawater. This system will be designed from previous experience building suspended sediment systems at James Cook University in Australia along with collaborating with the University of Hawaii. Once built we will be able to expose fish to varying levels of suspended sediments. This system will allow us to simulate observed suspended sediment events seen on coral reefs and predicted extreme events. Following exposure, we will test the physiology (aerobic scope, blood chemistry, swimming ability) and behaviour (anxiety, activity, burst response) of these fish to better understand how they will be impacted by suspended sediment levels (Figure B4.1). By conducting these experiments in parallel with the university of Hawaii and James Cook University we will be able to gain a robust understanding of how coral reef fish around the world are behaviourally and physiologically impacted by elevated suspended sediment events.
How the duration of suspended sediment events factors into the impact on marine organisms is that we know very little about. Marine habitats can go through varying durations of elevated suspended sediment where events can be acute, lasting days to weeks (Larcombe et al. 1995; Fabricius et al. 2013) or chronic, which can last weeks to months (Fabricius et al. 2014; Butler et al. 2015). During wet season these events can also occur in pluses where rain events increase suspended sediments for a shorter period of time but habitats are able to clear between rains (Field et al. 2007; Fong et al. 2020). Additionally, as climate change advances we are expected to see more frequent and intense storm events (Konisky et al. 2016) which would in turn increase runoff events. To understand how the duration of suspended sediment events impact the physiology of coral reef fish we intend to exposure fish to four treatments (Figure B4.2). Building from the previous experiment we will focus on the most sensitive physiological and behavioural metrics to access how they are influenced by duration of events. The results from this experiment help us understand how the duration of suspended sediments factors into the health of coral reef fish and will better allow us to manage our valuable marine environments.
3.9 Effect of microplastic on tuna meat quality and its intestinal microbiomes
This project was initiated upon a direct request by the Itoman tuna fishery association because they are concerned about the level of microplastic in the tuna they catch. My unit started this project in 2023 in collaboration with Prof. Cristine Luscombe (pi-Conjugated Polymers Unit) under the OIST’s mandate to promote Okinawa and its economy.
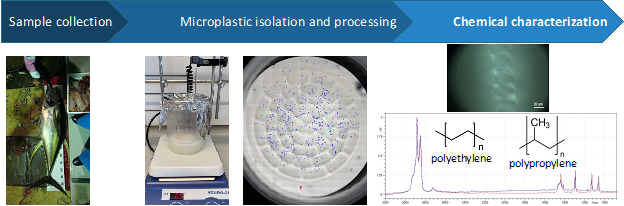
The presence of microplastics in the marine environment is an evolving phenomenon that can have widespread and lasting effects on marine organisms. Microplastic effects on aquatic organisms include impacts on reproduction, population dynamics, behavior, etc. These impacts can also have rippling effects on human populations that rely on marine food supplies. In particular, tuna are a major global food source and more than 7 million metric tons of tuna and tuna-like species are harvested yearly (“World Tuna Day 2 May” https://www.un.org/en/observances/tuna-day). Yet, despite the economic and social importance of the tuna industry, there have been no studies on the presence and abundance of microplastics in tuna. In this study, we investigate the presence, abundance, and chemical identification of microplastics in yellowfin tuna (T. albacares) gut samples in Okinawa, Japan. Currently, 10 gut samples have been processed according to the literature and have had microparticles (micro-sized particles before microplastic chemical confirmation) isolated on 45 nitrocellulose filter membranes (Martinelli et al., 2020). Figure 3.24 presents a summary of the workflow for microplastic analysis. All 45 nitrocellulose filter membranes have been mapped to generate filter maps and facilitate chemical analysis via Raman and FTIR microspectroscopy. The filter maps were then used to quantify and classify microparticles. A total of about 8,000 microparticles were located consisting of spheroid-, fragment-, and fiber-shaped particles. Based on preliminary results, we have identified microplastics as polyethylene (n=2), polyethylene/polypropylene copolymer (n=1), nylon (n=1), and graphitic material (n=2). The current goal is to perform chemical characterization on about 10% of the total number of microparticle shapes quantified.
The preliminary microbiome data showed that both tissues exhibit a similar bacterial richness, but the microbiome of the intestine is more evenly distributed (Figure A3.25a). In terms of overall diversity, there is a strong overlap between most intestine and stomach samples; however, there is a group of stomach samples that are strongly clustering separately (Figure A3.25b). There are some distinct differences in the microbial community, here shown at the phylum level (Figure A3.25c) and genus level (Figure A3.25d). In a next step, we plan to correlate these differences in alpha and beta diversity with the results of the microplastic analysis to investigate the potential effect microplastics can have on the tuna microbiome, and by extension tuna health.