Cell Signal Unit
Professor Tadashi Yamamoto
Abstract
The Cell Signal Unit studies molecular and cellular events that are relevant to and important for maintaining healthy life in a variety of environmental conditions. Through the studies, Unit explores the cause of various diseases that include cancer, neuronal disorder, immunological diseases, diabetes/obesity, and defects in development at the molecular level. The Unit characterizes regulation of gene expression, particularly post-transcriptional regulation that includes regulation by microRNA, and long non-coding RNA and RNA binding proteins. The Unit also characterizes protein kinase-mediated cell signaling especially in the control of the brain function such as emotions, learning and memory.
1. Staff
- Dr. Patrick Stoney, Staff Scientist
- Dr. Ken Matsuura, Staff Scientist
- Dr. Akiko Yanagiya, Staff Scientist
- Dr. Kristopher Paraiso Montrose, Postdoctoral Scholar
- Dr. Shou Soeda, Postdoctoral Scholar
- Dr. William Ashworth, Postdoctoral Scholar
- Dr. Olga Elisseeva, Visiting Researcher
- Ms. Saori Nishijima, Technical Staff
- Ms. Risa Ishida, Technical Staff
- Ms. Melissa Germany, Technical Staff
- Ms. Nao Ohmine, Technical Staff
- Ms. Atsuko Sato, Technical Staff
- Ms. Sandrine Anne Laure Burriel, Graduate Student
- Mr. Haytham Mohamed Aly Mohamed, Graduate Student
- Mr. Shohei Takaoka, Graduate Student
- Mr. Hemanta Sarmah, Graduate Student
- Ms. Dina Mostafa, Graduate Student
- Mr. Mohieldin Magdy Mahmoud Youssef, Graduate Student
- Mr. Hong Huat Hoh, Graduate Student
- Ms. Aisulu Maipas, Graduate Student
- Ms. Yuki Nakagawa, Research Unit Administrator
2. Collaborations
2.1 Physiology and Molecular Cellular Biology of the CCR4-NOT complex
- Description: Analyze the physiological and molecular biological roles of each component of the CCR4-NOT complex using gene-modified mice.
- Type of collaboration: Joint research
- Researchers:
- Dr. Keiji Kuba, Department of Physiology, Graduate School of Medicine, Akita University
- Dr. Toru Suzuki, Laboratory of Immunogenetics, Riken Center for Integrative Medical Sciences
- Dr. Toshinobu Fujiwara, Laboratory of Biochemistry, Faculty of Pharmacy, Kinki University
- Dr. Masahiro Morita, Department of Molecular Medicine, UT Health
- Dr. Nahum Sonenberg, Department of Biochemistry, McGill University
2.2 Basic cancer research for prevention and treatment
- Description: Search for substances that contribute to the prevention and treatment of cancer from biological resources, and elucidate its mechanism of action
- Type of collaboration: Joint research I and II
- Researchers:
- I. Representative director Kuniaki Nerome, Bioresource Laboratory LLC
- II. Professor Shinya Ikematsu, National Institute of Technology, Okinawa College
2.3 Mechanisms and physiological roles of CCR4-NOT complex-regulated gene expression in the brain
- Description: Electrophysiological studies of gene modified mice with abnormal social behavior
- Type of collaboration: Joint research
- Researchers:
- Team leader Masaru Tamura, RIKEN
2.4 Study of Cell-to-cell Interaction within Tumor Microenvironment Using An In-Vitro Three Dimensional Pancreatic Cancer Model
- Description:
- Establishing an in vitro 3D organoid culture for pancreatic ductal adenocarcinoma to study cell-cell interaction within tumor microenvironment
- Investigating the origin of cancer stem cells in pancreatic ductal adenocarcinoma by live cell imaging using tumor organoid
- Characterizing the effects of the interaction between stromal cells and cancer stem cells
- Identifying the targets for tumor-stroma crosstalk and cancer stem cells
- Type of collaboration: Joint research
- Researchers:
- I. Representative director Kuniaki Nerome, Bioresource Laboratory LLC
- Professor Masafumi Nakamura, Assistant Professor Kenoki Ohuchida, Dr. Kazuhiro Koikawa, Department of Surgery and Oncology, Graduate School of Medical Sciences, Kyushu University
2.5 Human immune responses and their modulation in cancer and autoimmunity
- Description: Human immune responses and their modulation in cancer and autoimmunity
- Type of collaboration: Joint research
- Researchers:
- Research Scientist Olga Elisseeva, Laboratory of Immunogenetics, Riken Center for Integrative Medical Sciences
3. Activities and Findings
3.1 Hepatic posttranscriptional network comprised of CCR4-NOT deadenylase and FGF21 maintains systemic metabolic homeostasis.
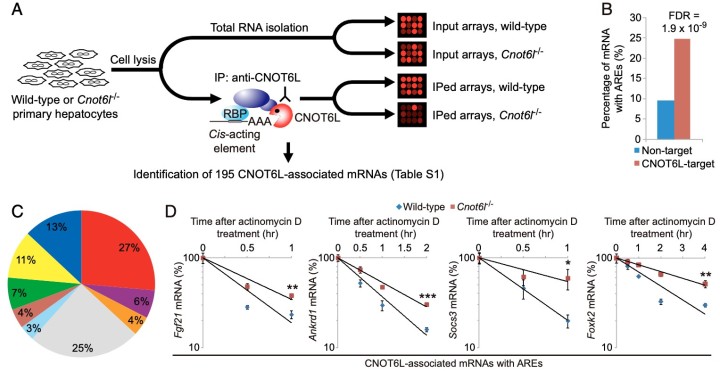
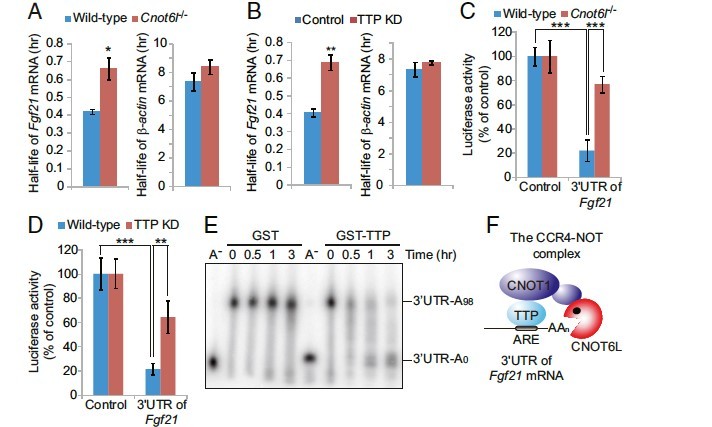
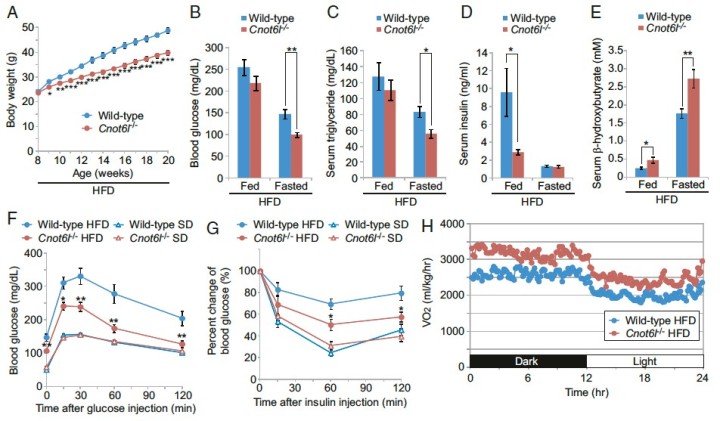
Whole-body metabolic homeostasis is tightly controlled by hormone- like factors with systemic or paracrine effects that are derived from nonendocrine organs, including adipose tissue (adipokines) and liver (hepatokines). Fibroblast growth factor 21 (FGF21) is a hormone-like protein, which is emerging as a major regulator of whole-body metabolism and has therapeutic potential for treating metabolic syndrome. However, the mechanisms that control FGF21 levels are not fully understood. Herein, we demonstrate that FGF21 production in the liver is regulated via a posttranscriptional network consisting of the CCR4–NOT deadenylase complex and RNA-binding protein tristetraprolin (TTP). In response to nutrient uptake, CCR4–iNOT cooperates with TTP to degrade AU-rich mRNAs that encode pivotal metabolic regulators, including FGF21. Disruption of CCR4– NOT activity in the liver, by deletion of the catalytic subunit CNOT6L, increases serum FGF21 levels, which ameliorates diet-induced meta- bolic disorders and enhances energy expenditure without disrupting bone homeostasis. Taken together, our study describes a hepatic CCR4–NOT/FGF21 axis as a hitherto unrecognized systemic regula- tor of metabolism and suggests that hepatic CCR4–NOT may serve as a target for devising therapeutic strategies in metabolic syndrome and related morbidities.
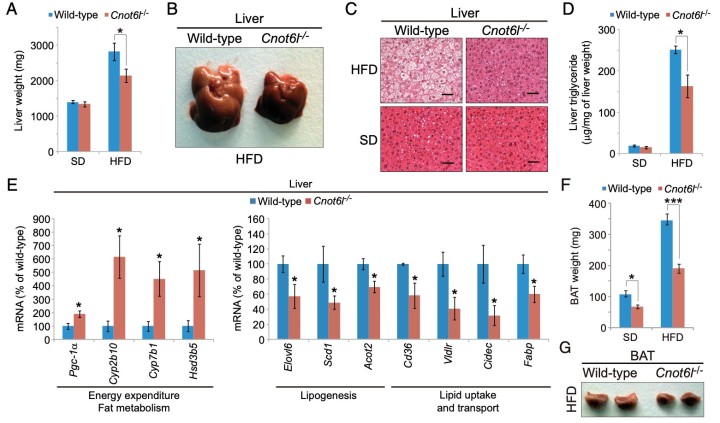
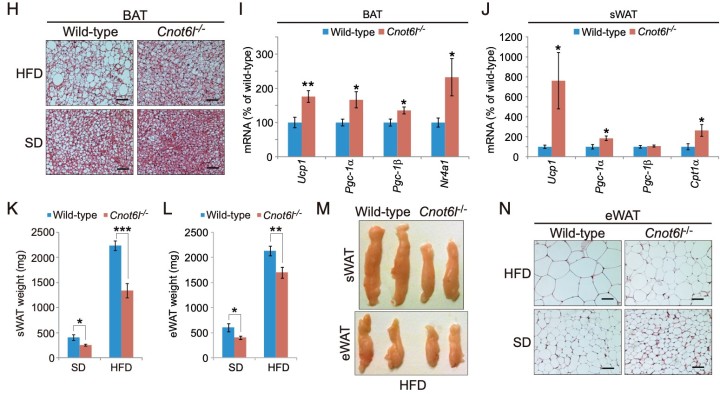
Figure 4. Cnot6l deletion leads to a resistance to lipid accumulation and an increase in energy expenditure in the liver, BAT, and WAT. (A) Weight, (B) representative image, (C) representative H&E staining, and (D) triglyceride contents of livers from wild-type and Cnot6l-/- mice fed on a standard diet (SD) or high fat diet (HFD). (Scale bars, 50 mm.) Liver triglyceride contents were normalized to liver weight. (E) Expression of genes involved in energy expenditure, fatty acid oxidation, lipogenesis, and lipid uptake and transport in the liver of wild-type and Cnot6l-/- mice fed on an HFD. Weight (F), representative image (G), representative H&E staining of BAT (H), and expression of the indicated mRNAs in BAT (I) and sWAT (J) of wild-type and Cnot6l-/- mice fed on an HFD. Weight of sWAT (K) and eWAT (L), representative sWAT and eWAT (M), and representative H&E staining of eWAT (N) from wild-type and Cnot6l-/- mice fed on SD or HFD. Scale bars in H and N, 50 mm.
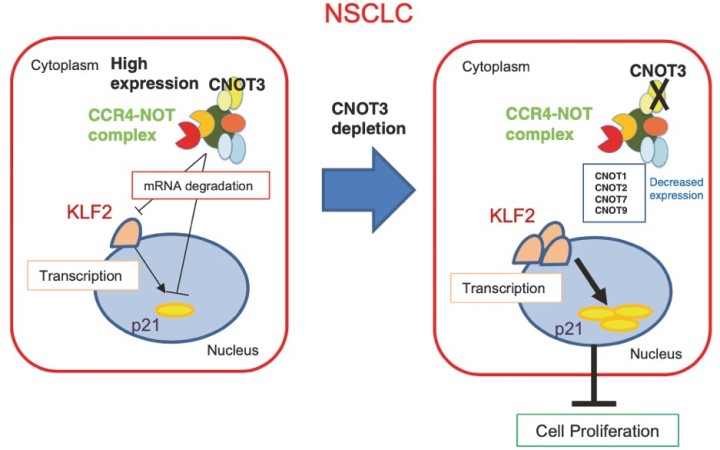
3.2 CNOT3 targets negative cell cycle regulators in non-small cell lung cancer development.
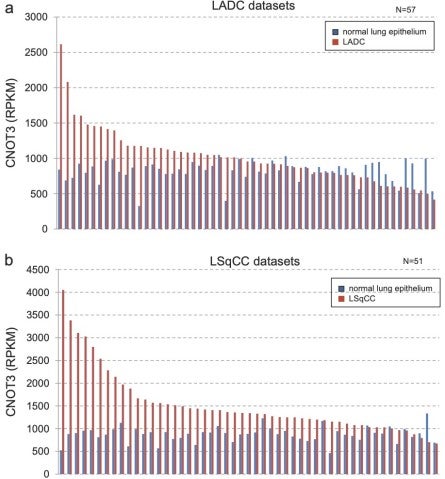
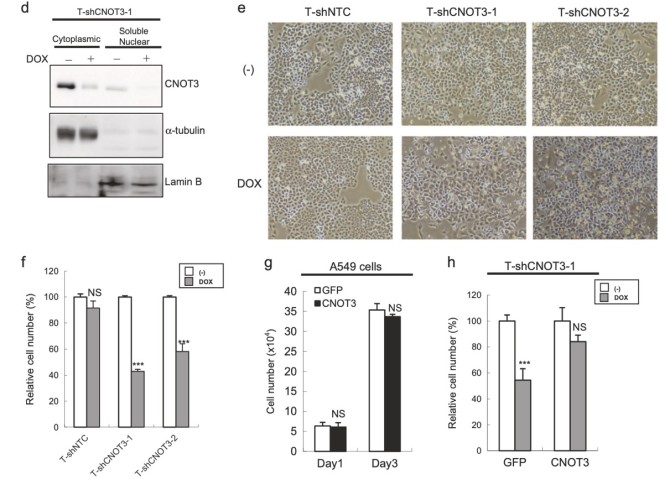
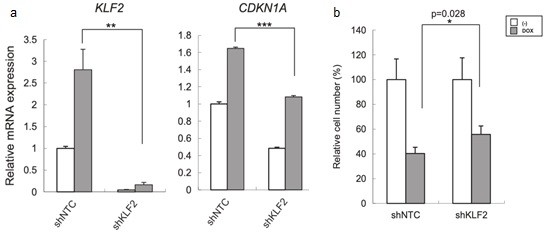
Lung cancer is one of the major causes of cancer death and clarification of its molecular pathology is highly prioritized. The physiological importance of mRNA degradation through the CCR4-NOT deadenylase has recently been highlighted. For example, mutation in CNOT3, a gene coding for CNOT3 subunit of the CCR4-NOT complex, is found to be associated with T-cell acute lymphoblastic leukemia, T-ALL, though its contribution to other cancers has not been reported. Here, we provide evidence suggesting that CNOT3 is required for the growth of non-small cell lung cancer. Depletion of CNOT3 suppresses proliferation of A549 human non-small cell lung cancer cells with enhanced mRNA stability and subsequent elevated expression of p21. In addition, we identified the mRNA for Krüppel-like factor 2 transcription factor, an inducer of p21, as a novel mRNA degradation target of CNOT3 in non-small cell lung cancer cells. Aberrant up-regulation of Krüppel-like factor 2 by CNOT3 depletion leads to impairment in the proliferation of A549 cells. Consistent with these findings, elevated mRNA expression of CNOT3 in non-small cell lung cancer in comparison with the paired normal lung epithelium was confirmed through scrutinization of the RNA-sequencing datasets from The Cancer Genome Atlas. Moreover, we found an inverse correlation between CNOT3 and CDKN1A (encoding p21) mRNA expression using the combined datasets of normal lung epithelium and non-small cell lung cancer. Thus, we propose that the up-regulation of CNOT3 facilitates the development of non-small cell lung cancer through down-regulation of Krüppel-like factor 2 and p21, contrary to tumor suppressive functions of CNOT3 in T-ALL.
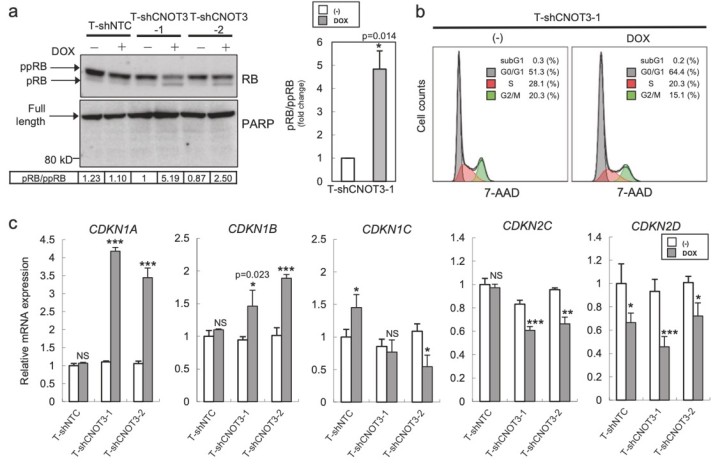
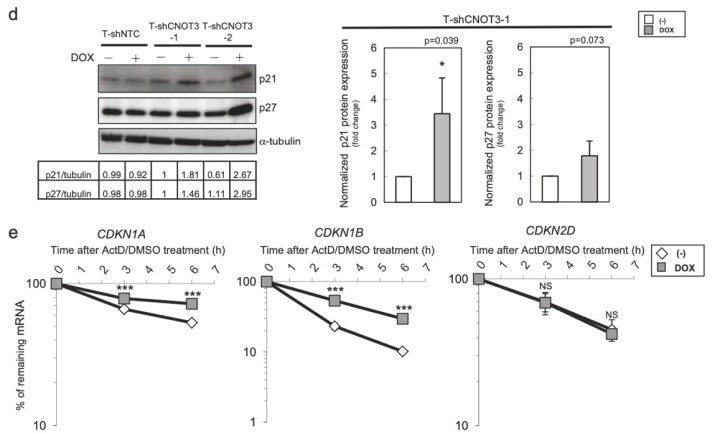
Figure 3. Depletion of CNOT3 induces the expression of p21 in human NSCLC cells. a, d Cell lysate of each A549 stable with or without 2 or 3 days of DOX treatment was subjected to immunoblotting with specific antibodies. ppRB, pRB, and full-length form of PARP are shown with arrows. Quantification of pRB/ppRB, p21 or p27/α-tubulin or β-actin was performed using Image J. The values were normalized to that of T-shCNOT3-1 without DOX. Average of the values obtained from four (p21 and p27) or three (pRB) independent sets of samples are shown. b Cell cycle analysis for A549-T-shCNOT3-1 cells with or without 3 days of DOX treatment. c qRT-PCR for CDKN1A, CDKN1B, CDKN1C, CDKN2C, and CDKN2D using the cDNA from the A549-T-shCNOT3-1 or -2 cells treated with or without DOX. e A549-T-shCNOT3-1 cells with or without 3 days of DOX treatment were treated with DMSO or ActD for 3 or 6 h. qRT-PCR for CDKN1A, CDKN1B, and CDKN2D using the cDNA from the cells with indicated time of treatment.
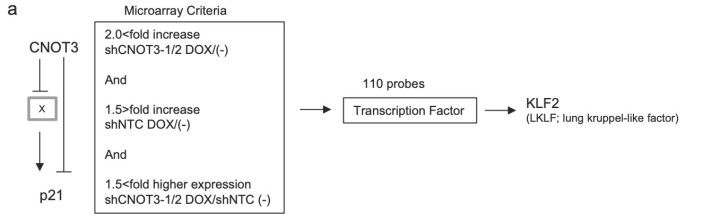
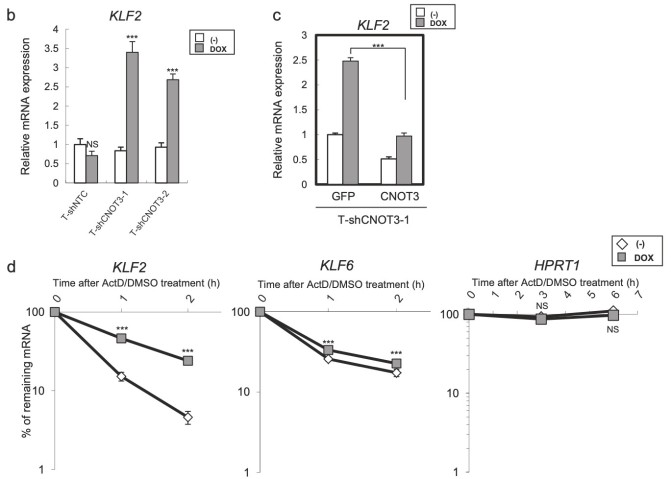
Figure 4. Identification of KLF2 transcription factor as a target of CNOT3. a Schematic model of regulation of p21 by CNOT3. We hypothesized that p21 expression is suppressed by a Factor X which is a target of CNOT3, in addition with direct mRNA decay by CNOT3. We narrowed down the candidate X to up-regulated 110 probes with microarray based on the criteria as follows: (1) More than 2-fold increase by both shCNOT3-1 and -2 induction. (2) Less than 1.5-fold increase by shNTC induction (to exclude non-specific increase). (3) More than 1.5-fold higher expression both in A549-T-shCNOT3-1 and -2 induced cells than A549-T-shNTC cells without DOX (to confirm the higher expression against A549-T-shNTC stable). We identified KLF2 as a Factor X. b qRT-PCR for KLF2 using the cDNA form the same samples with Fig. 1a. c qRT-PCR for KLF2 using the cDNA form A549-T-shCNOT3-1 cells expressing GFP or CNOT3 with or without 3 days of DOX treatment. d A549-T-shCNOT3-1 cells with or without 3 days of DOX treatment were treated with DMSO or ActD for 1 or 2 h. qRT-PCR for KLF2 and KLF6 using the cDNA from the cells with indicated time of treatment is shown. qRT-PCR for HPRT1 using the cDNA from the A549-T-shCNOT3-1 cells.
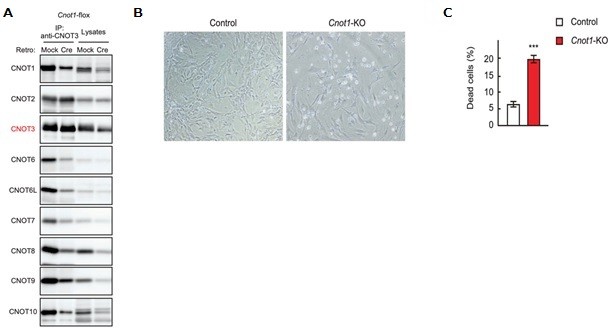
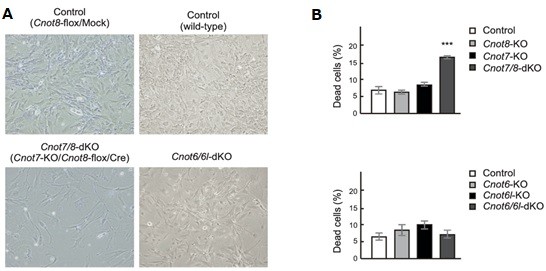
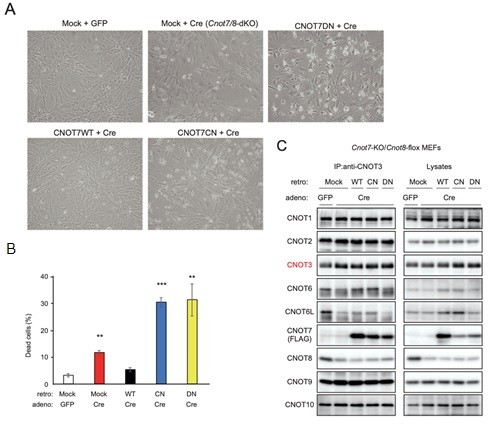
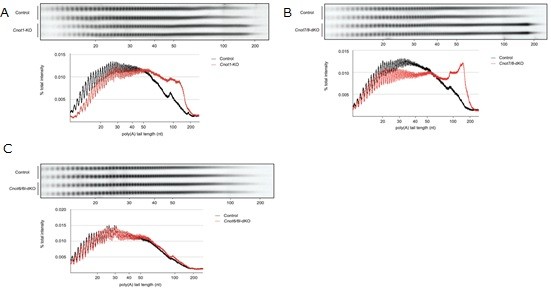
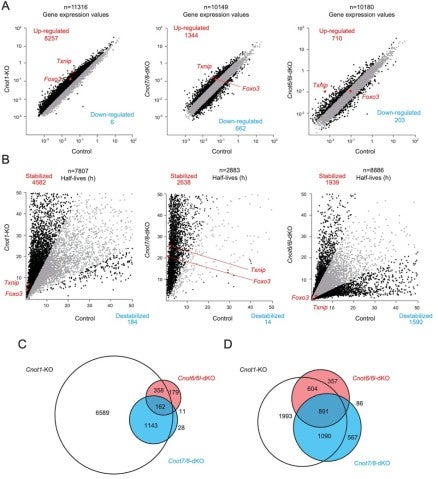
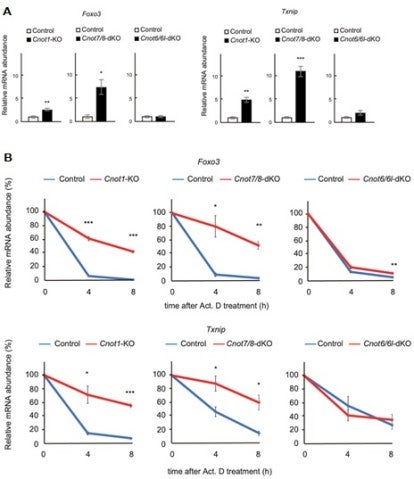
3.3 Essential functions of the CNOT7/8 catalytic subunits of the CCR4-NOT complex in mRNA regulation and cell viability.
Shortening of mRNA poly(A) tails (deadenylation) to trigger their decay is mediated mainly by the CCR4-NOT deadenylase complex. While four catalytic subunits (CNOT6, 6L 7, and 8) have been identified in the mammalian CCR4-NOT complex, their individual biological roles are not fully understood. In this study, we addressed the contribution of CNOT7/8 to viability of primary mouse embryonic fibroblasts (MEFs). We found that MEFs lacking CNOT7/8 expression [Cnot7/8-double knockout (dKO) MEFs] undergo cell death, whereas MEFs lacking CNOT6/6L expression (Cnot6/6l-dKO MEFs) remain viable. Co-immunoprecipitation analyses showed that CNOT6/6L are also absent from the CCR4-NOT complex in Cnot7/8-dKO MEFs. In contrast, either CNOT7 or CNOT8 still interacts with other subunits in the CCR4-NOT complex in Cnot6/6l-dKO MEFs. Exogenous expression of a CNOT7 mutant lacking catalytic activity in Cnot7/8-dKO MEFs cannot recover cell viability, even though CNOT6/6L exists to some extent in the CCR4-NOT complex, confirming that CNOT7/8 is essential for viability. Bulk poly(A) tail analysis revealed that mRNAs with longer poly(A) tails are more numerous in Cnot7/8-dKO MEFs than in Cnot6/6l-dKO MEFs. Consistent with elongated poly(A) tails, more mRNAs are upregulated and stabilized in Cnot7/8-dKO MEFs than in Cnot6/6l-dKO MEFs. Importantly, Cnot6/6l-dKO mice are viable and grow normally to adulthood. Taken together, the CNOT7/8 catalytic subunits are essential for deadenylation, which is necessary to maintain cell viability, whereas CNOT6/6L are not.
4. Publications
4.1 Journals
- Mostafa D, Takahashi A, Yanagiya A, Yamaguchi T, Abe T, Kureha T, Kuba K, Kanegae Y, Furuta Y, Yamamoto T, Suzuki T. Essential functions of the CNOT7/8 catalytic subunits of the CCR4-NOT complex in mRNA regulation and cell viability. RNA Biology, 17:403-416, 2020
- Nerome K, Ito-Kureha T, Paganini T, Fukuda T, Igarashi Y, Ashitomi H, Ikematsu S, Yamamoto T. Potent and broad anticancer activities of leaf extracts from Melia azedarach L. of the subtropical Okinawa islands. Am J Cancer Res, 10(2):581-594,2020
- Suzuki T, Kikuguchi C, Nishijima S, Yamamoto T. Insufficient liver maturation affects murine early postnatal hair cycle. Biochemical and Biophysical Research Communications 521:172-177, 2020
- Baba M, Yokoyama K, Seiriki K, Naka Y, Matsumura K, Kondo M, Yamamoto K, Hayashida M, Kasai A, Ago Y, Nagayasu K, Hayata-Tnaka A, Takahashi A, Yamaguchi S, Mori D, Ozaki N, Yamamoto T, Takuma K, Hashimoto R, Hashimoto H, Nakazawa T. Psychiatric-disorder-related behavioral phenotypes and cortical hyperactivity in a mouse model of 3q29 deletion syndrome. Neuropsycopharacology 44:2125-2135, 2019
- Takahashi A, Takaoka S, Kobori S, Yamaguchi T, Ferwati S, Kuba K, Yamamoto T, Suzuki T. The CCR4-NOT deanylase complex maintains adipocyte identity. Int. J. Mol. Sci. 20: 5274, 2019.
- Montrose K, Kobayashi S, Manabe T, Yamamoto T. Lmtk3-KO mice display a range of behavioural abnormalities and have an impairment in GluA1 trafficking. Neuroscience 414:154-167, 2019
- Ashworth W, Stoney P, Yamamoto T. States of Decay: The Systems Biology of mRNA Stability. Current Opinion in Systems Biology 15:48-57, 2019.
- Morita M, Siddiqui N, Katsumura S, Rouya C, Larsson O, Nakashima T, Hekmatnejad B, Takahashi A, Kiyonari H, Zang M, St-Arnaud R, Oike Y, Giguère V, Topisirovic I, Okada-Hatakeyama M, Yamamoto T, Sonenberg N. Hepatic post-transcriptional network comprising of CCR4-NOT deadenylase and FGF21 maintains systemic metabolic homeostasis. Proc Nat Acad Sci USA 116:7973-7981, 2019
- Shirai Y, Mizutani A, Nishijima S, Horie M, Kikuguchi C, Elisseeva O, Yamamoto T. CNOT3 targets negative cell cycle regulators in non-small cell lung cancer development. Oncogene 38:2580-2594, 2019
4.2 Books and other one-time publications
Nothing to report
4.3 Oral and Poster Presentations
- P. Stoney, A. Yanagiya, T. Yamamoto. Upregulation of Cnot8 in Cnot7 KO cells to maintain poly(A) homeostasis. The 42nd Annual Meeting of the Molecular Biology Society of Japan, Fukuoka, Japan, 2019.12.06 (2019)
- S. Nishijima, T. Suzuki, T. Yamamoto. Suppression of CNOT11, a subunit of CCR4-NOT complex, induces cell cycle arrest and autophagy. The 42nd Annual Meeting of the Molecular Biology Society of Japan, Fukuoka, Japan, 2019.12.06 (2019)
- W. Ashworth, Y. Yamamoto. Multi-omics analysis of mammalian CNOT4. The 42nd Annual Meeting of the Molecular Biology Society of Japan, Fukuoka, Japan, 2019.12.06 (2019)
- A. Yanagiya, T. Yamamoto. Post-transcriptional regulation in insulin biosynthesis by the Ccr4-Not deadenylase complex in mouse pancreatic β cells. The 42nd Annual Meeting of the Molecular Biology Society of Japan, Fukuoka, Japan, 2019.12.05 (2019)
- T. Yamamoto. Introduction to IMS and to my Research. Riken-KU Leuven Joint Symposium, KU-Leuven, Brussels, 2019.12.04 (2019)
- S. Soeda, T. Yamamoto. The role of maternal mRNA regulation by CCR4-NOT complex during mouse early development. The 42nd Annual Meeting of the Molecular Biology Society of Japan, Fukuoka, Japan, 2019.12.03 (2019)
- S. Takaoka, A. Takahashi, H. MA Mohamed, P. Stoney, T. Yamamoto. Brain-specific knock-out of 5’ - 3’ exoribonuclease Xrn1 causes hyperphagia and obesity. The 26th East Asia Joint Symposium on Biomedical Research, Seoul, South Korea, 2019.10.24 (2019)
- S. Soeda, T. Yamamoto. The role of gene expression regulation by CCR4-NOT mRNA deadenylation complex during mouse early development. RNA Frontier Meeting 2019, Shizuoka, Japan, 2019.09.24 (2019)
- M. Youssef, Y. Kiyama, H. Hamada, T. Suzuki, T. Manabe, T. Yamamoto. Transducer of ErbB2 (Tob) regulates stress in the brain. IBRO 2020, Daegu, South Korea, 2019.09.23 (2019)
- T. Yamamoto. Studies on breast cancer genes: history and latest trend. Okinawa Breast Cancer Research Association, Okinawa, Japan, 2019.08.24 (2019)
- K. Matsuura, T. Yamamoto. Investigation of the role of CNOT9 in mature brain. The 7th CCR4-NOT Meeting, Miyagi, Japan, 2019.07.27 (2019)
- P. Stoney, A. Yanagiya, T. Yamamoto. Upregulation of Cnot8 in Cnot7 KO cells to maintain poly(A) homeostasis. The 7th CCR4-NOT Meeting, Miyagi, Japan, 2019.07.27 (2019)
- H. Sarmah, T. Yamamoto. Role of mammalian CNOT9 in early embryonic development and differentiation. The 7th CCR4-NOT Meeting, Miyagi, Japan, 2019.07.27 (2019)
- S. Takaoka, A. Takahashi, H. MA Mohamed, P. Stoney, T. Yamamoto. Brain-specific knock-out of 5’ - 3’ exoribonuclease Xrn1 causes hyperphagia and obesity. The 7th CCR4-NOT Meeting, Miyagi, Japan, 2019.07.27 (2019)
- A. Yanagiya, T. Yamamoto. Post-transcriptional regulation maintains redox homeostasis in insulin biosynthesis by the Ccr4-Not deadenylase complex. The 7th CCR4-NOT Meeting, Miyagi, Japan, 2019.07.26 (2019)
- M. Youssef, Y. Kiyama, H. Hamada, K. Montrose, P. Stoney, T. Suzuki, T. Manabe, T. Role of Tob in the brain: An insight into stress-responsiveness function. Japanese Neuroscience society (Neuro2019), Niigata, Japan, 2019.07.25 (2019)
- A. Yanagiya, T. Yamamoto. Post-transcriptional regulation maintains redox homeostasis in insulin biosynthesis by the Ccr4-Not deadenylase complex. The 21st annual meeting of the RNA society of Japan, Tokyo, Japan, 2019.07.18 (2019)
- A. Yanagiya, T. Yamamoto. Post-transcriptional regulation to maintain redox homeostasis in insulin biosynthesis by the Ccr4-Not deadenylase complex in mouse pancreatic β-cells. mRNA Turnover Mechanisms, Regulation and their Implication in Infectious and Age-Related Diseases, Montreal, Canada, 2019.06.27 (2019)
- S. Soeda, K. Yamada-Nomoto, T. Michiue, M. Ohsugi. RSK-MASTL pathway delays meiotic exit in mouse zygotes to ensure paternal chromosome stability. The Annual Meeting of Japanese Society of Cell Science, Kobe, Japan, 2019.06.24 (2019)
- T. Yamamoto. Disease systems biology research in IMS and my cell signal study. RIKEN Center for Advanced Photonics, Riken, Wako, Japan, 2019.05.17 (2019)
- G. Vares, Y. Saintigny, F. Chevalier, C. Lepleux, M. Temelie, H. Hoh, V. Jallet, S. Sai, H. Sugawara, T. Nakajima. Targeting cancer stem cells with miRNA-based strategies and particle radiation therapy. HIMAC Annual Meeting, Chiba, Japan, 2019.04.22 (2019)
- G. Vares, Y. Saintigny, S. Sai, T. Nakajima. Characterizing and targeting cancer stem cells in challenging cancers. International Congress of Radiation Research (ICRR), Manchester, UK, 2019.08.26 (2019)
5. Intellectual Property Rights and Other Specific Achievements
5.1 Intellectual Property Rights
- Title: Anti-tumor agent
- Patent Number: JP6443872B2
- Inventors: Kuniaki Nerome, Tadashi Yamamoto, Taku Kureha, Reiko Nerome
- Owner: The Institute of Biological Resources
- Priority Date: Nov. 10, 2016
6. Meetings and Events
6.1 Seminar:
6.1.1 Heteromeric interference, a novel pathogenesis for human immunodeficiency
- Date: April 18, 2019
- Venue: OIST Campus Lab1
- Speaker: Dr. Ichiro Taniuchi (RIKEN, Yokohama)
6.1.2 Mechanisms determine gene silencing efficacies by miRNAs/siRNAs: thermodynamic properties and secondary structures.
- Date: May 8, 2019
- Venue: OIST Campus Lab1
- Speaker: Dr. Kumiko Ui-Tei (The University of Tokyo)
6.1.3 Decoding environmentally driven gene regulatory networks in hepatic macrophages
- Date: December 10, 2019
- Venue: OIST Campus Lab1
- Speaker: Dr. Mashito Sakai (The University of California, San Diego)
6.1.4 A hepatic post-transcriptional control of whole body metabolic homeostasis through FGF21 regulation by CCR4-NOT deadenylase
- Date: December 11, 2019
- Venue: OIST Campus Lab1
- Speaker: Dr. Sakie Katsumura (The University of Texas)
6.1.5 The reason why miRNAs bind to CDS
- Date: December 13, 2019
- Venue: OIST Campus Lab1
- Speaker: Dr. Hiroaki Sako (The University of Tokyo)
6.1.6 Portrait of 40-year-old p53
- Date: January 22, 2020
- Venue: OIST Campus Lab1
- Speaker: Dr. Rieko Ohki (National Cancer Center Research Institute)
6.1.7 Starting from the ends: from eukaryotic mRNA decay mechanisms to diseases
- Date: January 22, 2020
- Venue: OIST Campus Lab1
- Speaker: Dr. Bertrand Séraphin (University of Strasbourg)
6.1.8 The Not proteins regulate translation elongation dynamics
- Date: January 23, 2020
- Venue: OIST Campus Lab1
- Speaker: Dr. Martine Collart (The University of Geneva)
6.1.9 High throughput screening systems for inhibitors of phosphorylation dependent signaling molecules using fluorescent proteins
- Date: January 29, 2020
- Venue: OIST Campus Lab1
- Speaker: Dr. Nobumoto Watanabe (RIKEN, Wako)
6.1.10 How Aurora B activity is controlled at centromeres in mitosis
- Date: February 5, 2020
- Venue: OIST Campus Lab1
- Speaker: Dr. Toru Hirota (Cancer Institute of the Japanese Foundation for Cancer Research (JFCR))
6.1.11 Long non-coding RNAs required for MYC-driven cell proliferation
- Date: February 12, 2020
- Venue: OIST Campus Lab1
- Speaker: Dr. Peter K. Vogt (Scripps Research Institute)
6.1.12 Colon cancer metastasis: from mouse models to clinical applications
- Date: February 19, 2019
- Venue: OIST Campus Lab1
- Speaker: Dr. Makoto Taketo (Graduate School of Medicine, Kyoto University)
6.1.13 Transcription/export complex (TREX), elongation rate and mRNA 3´end processing
- Date: February 27, 2020
- Venue: OIST Campus Lab1
- Speaker: Dr. Teruko Tamura-Neimann (Hannover Medical School)
6.1.14 Cancer stem cells: targets for cancer eradication
- Date: Mar 18, 2020
- Venue: OIST Campus Lab1
- Speaker: Dr. Hideyuki Saya (Keio University School of Medicine)
6.1.15 Regulation of T cell activation and function by Innate signaling
- Date: Mar 25, 2020
- Venue: OIST Campus Lab1
- Speaker: Dr. Takashi Saito (RIKEN, Yokohama)
6.2 Meeting
6.2.1 ICSB 2019 (in collaboration with DoR)
- Date: November 1-5, 2019
- Venue: OIST Auditorium and Meeting Rooms
- Organizer (Core members): Shinya Kuroda (University of Tokyo), Miki Ebisuya (EMBL), Mariko Okada (Osaka University), Atsushi Mochizuki (Kyoto University), Katsuyuki Yugi (RIKEN), Kenichi Hironaka (University of Tokyo), Yohei Morita (OIST), Tomomi Nishi (OIST), Mayumi Kudo (JST), Haruka Ikuta (JST), Tadashi Yamamoto (OIST)
- The number of Participants: 442
6.2.2 The State-of-the-Art 3D Tissue Culture & Organoids Symposium (in collaboration with DoR)
- Date: April 18-20, 2019
- Venue: OIST Seaside House.
- Organizer: Keiko Kono (OIST), Mitsuru Morimoto (RIKEN), Toshiro Sato (Keio University), Minoru Takasato (RIKEN), Tadashi Yamamoto (OIST)
- The number of Participants: 30
7. Other
Nothing to report.